
These unexpected deaths of laboratory mice are caused by kidney disease, as these organs appear shrunken and fibrotic upon histologic examination, with tubular necrosis, interstitial fibrosis, and intranuclear inclusion bodies. Sequence analysis of RNA from diseased kidneys revealed a 4,442 nucleotide genome with homology to parvoviruses. This new agent was called mouse kidney parvovirus (MKPV).
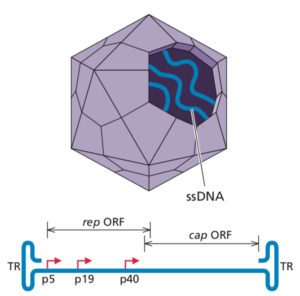
Parvoviruses (illustrated) are small viruses with single-stranded DNA genomes. They cause the human rash diseases erythema infectiosum (fifth disease) and arthritis, and serious infections in dogs and cats. There are also parvoviruses that infect mice, but these do not cause disease. MKPV is very different from these parvoviruses; it is more related to isolates from Old World fruit bats and vampire bats and are likely part of a new genus within the family. Whether MKPV is present in mice in the wild is unknown.
Co-housing experiments show that MKPV is transmitted among mice via feces and urine. After transmission the virus can be detected in serum and urine within 50-80 days, after which mice lose weight and die from kidney disease. The virus replicates within tubular epithelial cells in the kidney, and the abundance of viral genome sequences correlates with histological disease severity. Disease is mainly observed in immunodeficient mice, in which virus replicates to ten fold higher levels than in immunocompetent mice.
Chronic kidney disease in humans may have a variety of causes, including diabetes mellitus, hypertension, and virus infections. The latter are problematic in organ transplant recipients, in which polyomavirus-associated nephropathy is a major cause of death. The severity of interstitial fibrosis is a determinant of renal failure, but measuring fibrosis in patients is difficult. Several urine and serum biomarkers have been identified and some are found in mice infected with MKPV. Infection of mice with MKPV might therefore be a useful model for studying kidney interstitial fibrosis.
The authors conclude that MKPV is the cause of inclusion body nephopathy in mice. But Koch would be turning over in his grave: the authors did not fulfill his postulates, because they did not isolate MKPV, propagate it in culture, reintroduce it into mice, show that mice developed the same disease, and reisolate the virus from these diseased mice.
However, we are in a new age far beyond Koch where many viruses are discovered in silico (as was MKPV) and cannot be propagated in culture. Indeed, the authors indicate that they were unable to grow MKPV. A new set of Koch€™s postulates are required for this new age, as outlined by Fredericks and Relman and previously discussed on this blog. Authors claim they have fulfilled most of these new requirements:
- MKPV was found only in diseased mice, and mainly in kidneys, the only organ to display disease.
- MKPV viremia was detected before disease, and viral DNA copy number correlated with disease.
- Parvoviruses assemble in the cell nucleus, consistent with the observation of inclusion bodies in the kidney epithelium.
- Viral sequences were detected in affected but not normal cells of the kidney
It seems likely that MKPV causes IBN in mice. Nevertheless, I am certain that the authors continue to try to propagate MKPV in cell culture, so they will one day allow Koch to remain at rest in his grave.

Pingback: Leaving Koch behind – Virology