
Acute lymphoblastic leukemia, or ALL, is caused by uncontrolled growth of B cells, which normally produce antibodies to fight off infections. It is the most common cancer in children. The uncontrolled production of these cells by the bone marrow causes a shortage of blood cell production, leading to fever, increased risk of infection, and anemia. These B cells have on their surfaces a protein called B19 – which is the key to understanding how Kymriah works.
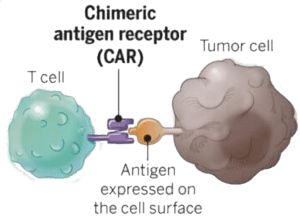
The therapy begins with drawing blood from the patient, from which T cells are purified. These T cells are then infected with a lentivirus vector that encodes the gene for a chimeric antigen receptor (CAR) that recognizes the B19 protein. The CAR protein is synthetic – it doesn’t exist in any cell. The extracellular domain consists of a single-chain antibody directed against the B19 protein (pictured). The cytoplasmic domain of the protein contains sequences that stimulate the T cells to proliferate.
After the T cells are infected with the CAR-encoding lentivirus, they are infused back into the patient. Upon encountering a B cell producing B19, the T cells bind to the protein and kill the cells, thus eliminating the cancer.
Kymriah was licensed by the FDA after testing showed it was effective, leading to remission of cancers in the majority of children treated. But the price tag is steep – $475,000 for a treatment, and other similar drugs in the pipeline could be even more expensive. The drug makers justify the high price by arguing that it reflects the value to the patient – it saves their lives.
But vaccines also save lives, and they cost much less than Kymriah. The difference, of course, is that vaccines are given to millions of people. Kymriah, in contrast, would be given to thousands in the US.
In other words, the high cost of Kymriah reflects the need of drug companies to recoup their high investment in developing and testing the drug – not the value to the patient. Rather than spinning a false story about the value of a drug to a patient, the drug companies should be honest about the pricing of their products. No wonder the public has a negative image of the industry.

Pingback: The $475,000 drug - VETMEDICS
A fine example of what happens on the rare occasion (unique?) when a drug company actually manages to produce something that cures: instead of the usual practice of creating cash cow drugs that have to be taken for life.
This is brilliant idea, but, because it works, ‘customers’ won’t have to keep coming back for more. Few drug cos will want to find themselves in this situation!
A glaring example of why we need public pharmaceutical services that both research real *cures* and are funded to produce the ‘single use’ substances that could actually solve diseases for the universal public good.
I’m grateful that they’ve worked to create this (my stepdad died a few years ago from blood cancer, and I only wish I knew about this; his chemo brought the cancer down until it was almost gone, but by the time the chemo was stopped, the cancer immediately started growing again, and overtook him).
However, more needs to be done about making these things more affordable!
Then again, any funding efforts would probably be corrupted somewhere down the line, in order to serve the selfish interests of certain others (kind of like a virus hijacking a cell’s genes).