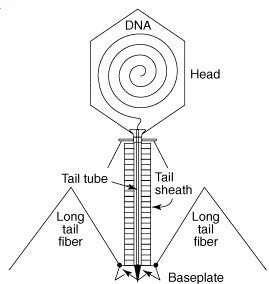

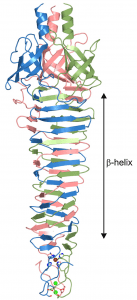

Structures of the spike from P2, a well studied virus of E. coli, and the choleraphage phi92 were determined. The spikes are built from three copies of a single protein (trimers). The trimers are indeed shaped like spikes: they are wider at one end and taper to a rather sharp tip (figure at right). The bulk of the spike is made up of alternating beta-strands which form a corkscrew-like beta-helix. The sharp tip is composed of three beta-hairpins which the authors say “come together like petals in a flower bud”.

To verify that these structures are those of the spike that is attached to the baseplate, the authors solved the structure of the phage particle by cryo-electron microscopy and image reconstruction. The image clearly shows the spike protruding from the base plate (figure below). The structures of the spike proteins solved by X-ray crystallography could then be computationally fitted in the correct location in the cryo-EM image of the baseplate.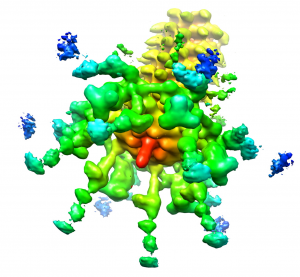

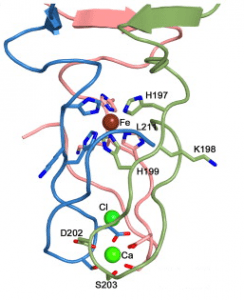
These structures support the idea that the spike is a rigid needle that pierces the bacterial membrane and forms a channel through which the DNA can pass. An interesting question is how the DNA gets past the spike, which plugs the end of the tail tube. The authors believe that the spike is loosely attached to the tube and might be easily dissociated once it passes through the cell membrane. The spike of phage T4 can be dissociated at low pH, a condition that is found in the periplasm, the space between the inner and outer bacterial membranes.
There are distinct signatures of the spike structure that can be identified in proteins of other contractile injection systems, including diverse bacteriophages. They can also be found in bacterial type VI secretion systems, which are membrane complexes used to transport proteins outside of the cell. Once evolution builds a useful machine, it is often put to many diverse uses.
Browning, C., Shneider, M., Bowman, V., Schwarzer, D., & Leiman, P. (2012). Phage Pierces the Host Cell Membrane with the Iron-Loaded Spike Structure, 20 (2), 326-339 DOI: 10.1016/j.str.2011.12.009

I have to admit, this is maybe one of the coolest things i`ve ever seen. I always wondered about how Phages pierce the bacterial cell membrane, but seeing how the model of them using the spike is translated into a three-dimensional structure that even looks somehwat like a drill or a needle is just… fascinating. It also makes one just stand back in awe about how nature evolved something so beautiful that is yet such an effective molecular machinery. Thanks a LOT for providing this story, since my university doesn`t supply access to Structure i would not have had a chance to take a look at it.
This story amazes me… it actually looks like a little molecular drill! Perhaps this is a naive question, but you mention that the iron atom held at the center of the spike adds strength to the structure, and would aid it in piercing the cell membrane. Do single atoms of metals such as iron have the macro properties of the substance such as “hardness”, or by strength do you mean increased structural stability due to strong attractions between the histidine residues and the iron atom?
The amount of pressure all controlled by proteins, nucleic acid, some coordinated iron and the biophysical beauty of this system amazes me!
I would suppose the iron stabilizes the 3D structure of the trimer and the binding of each monomer to the complex.
 I havent read the paper yet, but the other way would be a lot into alchemy.
Correct, the iron stabilizes the trimer because it interacts with histidines from all three subunits.
Dr Rachaniello. Could you clear something up.
If there is no env region in the virus that is in 22Rv1. Then doesn’t that mean there is no contamination?Â
This explains it.
http://www.me-advocacy.com/Missing_22Rv1_env_region.html
Pingback: How do Viruses Reproduce? – Immunology