
Perhaps this is a place to bring up particle to pfu ratio? The above is great for when talking about phage, for example, when the ratio approaches 1. But with something like polio when it can be very high (>1000 ??), then it’s not that all cells don’t receive “a particle” at MOI=1 – but that they don’t get an “infectious dose”. Not sure how to say it better – enough to initiate an infection.
So why does polio require 1000 virions to make an infectious dose? I don’t buy the idea that most of the particles are not “viable”.
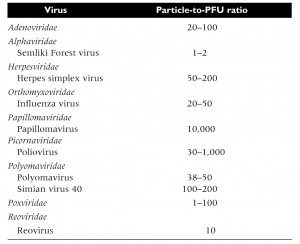
If we take the titer of a virus preparation (in plaque forming-units per milliliter) and divide it by into the number of virus particles in the sample, we obtain a number known as the particle-to-PFU ratio. It is a measure of the fraction of virus particles in a given sample that can complete an infectious cycle. For many bacteriophages, the particle-to-PFU ratio approaches 1, which is the lowest value that can be obtained. A value of 1 means that every virus particle in the sample is able to form a plaque.
For animal viruses, the particle-to-pfu ratio is often much higher, from 1 to 10,000 (the image shows values for different animal viruses – click to enlarge). These high values complicate the study of animal viruses. When the particle-to-pfu ratio is high, one can never be certain that properties measured in infected cells are those of the infectious or the non-infectious viral particles.
The linear nature of the dose-response curve indicates that a single virion is capable of initiating an infection. However, the high particle-to-pfu ratio of many viruses shows that not all virions are successful. A high particle-to-pfu ratio is sometimes caused by the presence of noninfectious particles with genomes that harbor lethal mutations or that have been damaged during growth or purification. Another explanation is that although all viruses in a preparation are in fact capable of initiating infection, not all of them succeed because of the complexity of the infectious cycle. Failure at any one step in the cycle prevents completion.
A high particle-to-pfu ratio does not indicate that most particles are defective, but that they failed to complete the infection.

Pingback: Tweets that mention Are all virus particles infectious? -- Topsy.com
Should the first sentence of the 2nd paragraph read?: “If we take the titer of a virus preparation (in plaque forming-units per milliliter) and divide it INTO the number of virus particles in the sample, we obtain a number known as the particle-to-PFU ratio”
You are correct. I’ll fix it.
Why don’t my comments appear?
Your comment was caught in the spam queue. It should appear shortly.
OK, for a virus like polio (+sense ssRNA) to generate progeny virus, 1) the infecting virus has to release a “competent genome” (full length, no debilitating mutations); 2) the genomic RNA has to “connect” with a ribosome and be completely translated – perhaps multiple times; 3) the resulting polyprotein has to undergo the correct self-cleavages (is this more efficient in trans rather than cis?); 4) the replication complex then has to generate a template (-sense ssRNA) before the genome is degraded; 5) the replication complex has to then generate new genomic RNA before the template is degraded.
I would have guessed that upon initial infection all these steps would have a probability of success considerably lower than 1 due to concentration effects.
Transcription and translation of almost 10kb takes quite a while – and this length is about 10 x the average eukaryotic mRNA. I would also expect eukaryotic translation rates have evolved to favor the “average message”, perhaps at the expense of error/success rates.
All in all, I think that if single polio genome enters a cell, it has a very low probability of successfully generating progeny virus.
I just thought I’d add my 2 ha’porth (or cents’ worth, for you in the US) here, and point out that some viruses do need at least two particles infecting the cell in order to successfully start an infection. These are viruses with “multipartite” genomes, in which each physical particle only contains part of the virus genome. There are several virus families like this in plants, though as far as I know no animal viruses with multipartite.
Am I right in thinking that this is linked to the fact that many plant viruses are transmitted by insect vectors? This would make sense, because an insect that dined on an infected plant may take up a lot of virus particles, that would all be injected together when the insect goes on to infect another plant.
Anyway, I guess this requirement for “two hits” (two particles infecting a single cell) shows up on virus titre assays like the pfu test, right?
DMc
Very interesting notes! Assuming perfect attachment and entry conditions are achieved, an infectious particle must have minimal number of lesions in its genome to replicate in the cell. In this respect, RNA viruses may have high physical particle to infectious particle ratios since these viruses exist as quasi species due to RNA-dependent RNA polymerase associated mistakes (as many as 10^-3 – 10^-5 mutations per nucleotide per replication event). This kind of argument may also apply to segmented viruses that have packaging issues. You raised a very good point about the compatibility of the eukaryotic translation [and probably transcription] machineries. Besides, the replicating virus is under pressure from antiviral factors produced by the infected cell and its neighbors.
Isn’t this how some hepatitis viruses work?
Well, hepatitis delta does need hepatitis B virus (HBV) coinfection, but that’s a satellite virus rather than an integral part of HBV. During blood transfusion, a lot of HBV and hep delta particles can be transferred together, which I guess favours co-infection. I have no idea how hepatitis delta propagates in a more natural setting. Perhaps during mother-to-child transmission, since this is a major mode of HBV infection.
Come to think of it satellite viruses are pretty frequent in plant viruses too, although only
So for these multipartite genomes, is the splitting of the genome into two particles obligate? Do any of the particles contain the entire genome? Or is the sorting of the parts of the genome into different virus particles stochastic.
Yes, it is obligatory.
No, as far as I know, no particles contain the whole genome.
I don’t know, but I would guess that it is.
Some plant viruses split their genome into three particles, not just two – like hordeiviruses
http://www.expasy.ch/viralzone/all_by_species/42.html
The question begs the question: What is meant by the word “infectious”? Even in the above post, several meanings are implied in using the word “infectious” or “infect” or “infection” and lead to modifiers and qualifiers, for example, “infectious cycle”, “infectious dose”, etc. Then, there is “successful infection”. How do we define “infect” when we are referring to virus/cell encounters.
PFU, of course, only applies to those viruses that are, in fact, capable of forming plaques in cell cultures. If a virus particle enters a cell, makes some RNA or DNA and maybe some proteins, but doesn’t kill the cell or make any progeny, is that cell “infected”?
Infectious is a fuzzy word! Its use leads to fuzzy questions! If a tree fell in the forest and no one was around to hear it, did it make a sound?
It’s a good teaching concept: Critical thinking vs. Fuzzy logic. We all know what “infectious” means, don’t we?
I define infectious to mean completion of the replication cycle, from
entry to synthesis of new particles. In this example it specifically
means PFU. The title of the post should be ‘can all viral particles
form plaques’, but in fact many viruses cannot form plaques on any
cells. Others use infectious in other ways – but I don’t believe it is
correct. The term ‘abortive infection’ is instructive – the virus gets
in but stops at some point before making new particles. This implies a
complete cycle for infection. But I agree, it is a good teaching
concept – in fact in virology if you examine the terms you can learn a
lot.
Hi Professor, very helpful table and discussion. Do you have any citations for the particle-to-PFU ratios, such as that for influenza virus? This information tends to be buried in the classic literature and textbooks and is not so easy to find in pubmed searches.
With the caveat that “an excellent analogy is a poor explanation”, perhaps viewing this as an analogy of an exothermic chemical reaction with an energy of activation might be useful. In the analogy all molecules are capable of undergoing the reaction to produce products and energy. However, in order for the reaction to occur, an initial input of energy is required to get over the energy of activation hump. Once that happens, all molecules react via the production of energy from other molecules reacting. Net result: a reaction occurs for each molecule.
In this analogy, different chemical reactions have different E(act): some less, some more. Viruses with a low particle to pfu ratio (or whatever), require less of a “push” to make the “infection” work.
What is the E(act) for a virus infection? Perhaps cis-acting functions in the virus replication cycle are less efficient than trans-acting functions for some viruses. In this analogy, multiple particles initiating a replication cycle in a single cell might provide the trans-acting biological “energy” to overcome the “probability hump” to get the replication cycle going.
Poliovirus is a good case to look at. Initiation of infection by a single particle depends on translation of the +RNA, self-cleavage of the polyprotein, processing to functional polioviral enzymes, and amplification of the RNA. The poliovirus +RNA is handicapped (handi-uncapped?) intially; a single +RNA (handi-uncapped) must compete with millions of cellular mRNA’s for translation. Although a single polioviral +RNA might be sufficient to initiate an infectious cycle, the probability of it being translated in the presence of millions of mRNA’s with a cap advantage is very low.
Boost the number of poliovirus +RNA’s in the cell a thousand-fold and, boom!, the probability of successful translation is increased (a thousand-fold). Once that happens, it is all downhill for the cell and uphill for poliovirus! The poliovirus protease knocks out the capped mRNA advantage and polio RNA takes over the cell machinery. One poliovirus RNA (particle) is sufficient but many are necessary to overcome the initial “E(act) hump”.
Sorry, just my late night musing…
It is important to point out that if the number of physical particles is significantly higher than the infectious titer another explanation may be that the assays used to measure titer do not count every infectious particle. In our experience with retroviruses we often hear that only 1 in 1000 particles are infectious, but such assertions do not generally have any citations as everyone assumes this is true. Crawford and Crawford (1961, Virology 13:227) reported that for RSV the ratio of physical particles to infectious particles was 750 to 1; however, they felt that this may have been because “only a small proportion of the virus particles may absorb to the cells, owing to the sparse cell layer used in the RSV assay.†In our studies of HIV-1 we proposed that the biggest hurdle to an infection is for a virus to encounter a cell (Thomas et al 2007 JVI 81:4367). This hypothesis explains why in HIV-1 cell-cell transmission is far more efficient than cell-free transmission (Lekkerkerker 2006 Curr HIV Res 4:169; Piquet and Sattentau 2004 J Clin Invest 114:605). Several other groups have published reports that expand on this idea (Platt et al 2005 JVI 79:4347; Munch et al 2007 Cell 131:1059; Roan et al 2009 JVI 83:73; Brender et al 2009 Biophys J 97:2474).
In your JVI paper you speculate that “The true ratio of infectious to defective particles is likely several orders of magnitude higher, i.e., 1 in 8 to 1 in 20 rather than 1 in 1,000 to 1 in 60,000” meaning that, in your view, noninfectious particles do exist. One possibility for the existence of noninfectious particles is the failure to incorporate the genome into the nascent virions. For example, it has been possible to make empty virus-like particles using the influenza virus proteins that associate with the envelop. Are there data suggesting that all HIV virions have the genome?
This is an interesting question. What kind of laboratory experiment would support of refute Dr. R’s hypothesis that polio’s high particle/pfu results more from abortive infection rather than damaged, non-infectious particles? Perhaps this could be addressed by measuring this ratio in different cell lines in vitro using the same stock preparation of virus. Cells that are more or less susceptible to infection may display correspondingly lower or higher particle-to-PFU ratios depending on the rate of abortive infection, which would support Dr. R’s hypothesis. However, if the ratios are consistent between cell lines (same #’s of plaques, but of different sizes depending on permissivity), this would refute his hypothesis.
This begs the question of how the data presented in the table were arrived at. Note that the value for PV is not 1000, but rather 30-1000 which is a wide range. Understanding the basis for this range in particle-to-pfu ratios may shed light on this question.
Noninfectious particles can manifest themselves easily if they have measurable biological activities. A good example is the classical Defective-interfering particles (DIP) described by von Magnus in 1954 [Adv. Virus Res. 2:59-78]. Thus, DIP titer would be different from infectious particle titer in a stock with mixed subpopulations when measured in the same type of cells. Note: Noninfectious particle (DIP in this case) titers are statistically calculated using the Poisson Distribution given a measurable dose-dependent biological activity (Plaque Yield reduction in this case).
Hi, could you mention the source of the table that you have used in your post? Thanks
The table is from Principles of Virology, Flint et al., Third Edition,
ASM Press.
great ….information for my knowledge
http://www.biotechview.blogspot.com
If I supose that the particle to PFU ratio is 10000:1, and if I use 10000 particles to infect a monolayer with 1000000 cells, this could be, that at least in 100 cells one could be infected by more than one particle and form that single plaque? taking into account that the M.O.I is o.01?
If you consider only the infectious viruses then the MOI is 0.000001 (you infect one million cells with one pfu). But if you consider all particles the MOI is 10,000/1,000,000 or 0.01. At that moi the fraction of cells that will receive more than one virus particle is 0.01 percent – in a culture of 1 million cells that amounts to 100 cells. Whether or not infection of a single cell with more than one virus will lead to complementation of noninfectious virions depends on how many defects exist in the viral population. It is theoretically possible but highly unlikely.
Pingback: The Lazarus virus
Pingback: The Lazarus virus | StigmaBot
Sir ,Â
I would like to know about Particle senses disease pls help me out …
 adis
Infectivity is 2x infection(entry)
 Hi.. May i point out a paper which micr-injected FMDV RNA and found the PFU / RNA molecule to be approaching 1.
Yes, Please point out.
Is there a reference for the phage particle infection efficiency of nearly 1? I’m looking for the efficiency of T7 type phages, would be great if someone can point me to a literature because i could not find ANY after hours of search. Thanks!
I know this is an old thread, but I was searching through literature for studies that looked for defective particlesduring acute infection of an animal (sorry, I am biased). Came across the below paper claiming to be the first study showing DI particles during an acute infection… I am a teacher and don’t have the time to ferret out the literature completely, but figure that others looking at this thread might find it useful. Below that link is a second paper citing the first that finds defective particles in WNV infections of parakeets.
http://www.plosone.org/article/info%3Adoi%2F10.1371%2Fjournal.pone.0019447
http://www.ncbi.nlm.nih.gov/pmc/articles/PMC3312038/
Pingback: What do you mean by ‘infectivity’? A conversation with Richard Hardy | MRC-University of Glasgow Centre for Virus Research science blog