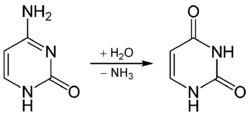
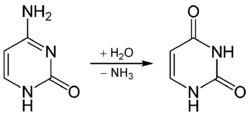
During retroviral replication, Apobec proteins are packaged into newly synthesized retrovirus particles (illustrated). 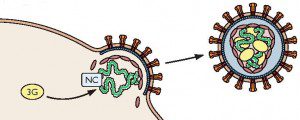

Apobec is lethal for retroviruses that incorporate the enzyme into their virions. Human immunodeficiency virus-1 counters this defense by producing the Vif protein, which binds to Apobec and promotes its degradation by cellular enzymes.
XMRV does not encode a Vif protein and should be susceptible to inhibition by Apobec proteins. To answer this question, XMRV virions were produced in cells in the presence of different Apobec proteins. The deaminases were incorporated into virions, where they resulted in G-to-A hypermutation and inhibition of viral infectivity.
Could the presence of Apobec determine which human tissues are infected with XMRV? The virus replicates very well in a prostate cancer cell line, LNCaP, which produces reduced levels of Apobec proteins. Whether Apobec could regulate XMRV replication in the prostate is not known because expression of the protein in normal or malignant prostate tissues has not been studied. A conundrum which requires further investigation concerns the isolation of XMRV from CD4+ T and B cells, which are known to synthesize Apobec proteins. How XMRV might evade Apobec inhibition in these cells remains unexplained.
Paprotka, T., Venkatachari, N., Chaipan, C., Burdick, R., Delviks-Frankenberry, K., Hu, W., & Pathak, V. (2010). Inhibition of Xenotropic Murine Leukemia Virus-Related Virus by APOBEC3 Proteins and Antiviral Drugs Journal of Virology DOI: 10.1128/JVI.00134-10
Groom, H., Yap, M., Galao, R., Neil, S., & Bishop, K. (2010). Susceptibility of xenotropic murine leukemia virus-related virus (XMRV) to retroviral restriction factors Proceedings of the National Academy of Sciences, 107 (11), 5166-5171 DOI: 10.1073/pnas.0913650107

Could enteroviruses have anything to do with it?
Nora Chapman and Stephen Tracy at the University of Nebraska have shown persistance of enteroviral infection in the apparent absence of infectious particles, and enteroviruses and CFS have a long association due to the epidemic nature of some outbreaks, incubation period, etc.
One of, if not the first known outbreak of CFS was documented by future U.S. Surgeon General Alexander Gilliam at the Los Angeles County Hospital in 1934, with Dr. Gilliam calling the illness either 'atypical' or 'abortive' poliomyelitis, I can't remember which, although both descriptors have been used at one time or another when describing outbreaks of CFS.
I think I've also read that the LACH staff was innoculated with serum from polio survivors, is it possible that one of the individuals serum was taken from was infected with XMRV?
Also there's something wrong with PWC's RNaseL system, which produces a truncated 37kda form not found in healthy controls.
Goody goody goody, time for people to start putting all the pieces of the puzzle together!
“How does a lytic enterovirus persist and cause chronic disease?” – Nora Chapman
International Symposium on Viruses in CFS & Post-viral Fatigue
A satellite meeting of the 6th International Conference on HHV-6 & 7 in Baltimore, Maryland, USA Day 4 – June 23, 2008 – Enterovirus Session
http://www.scivee.tv/node/7031
'Human Enteroviruses and Chronic Infectious Disease' written for (or at least (re)published in) ME/CFS charity Invest in ME's newsletter Journal of IiME.
http://www.investinme.org/Documents/Journals/Jo… -p.23
I think it would be very useful to conduct viral discovery on samples
from CFS patients, to discover whether any other pathogens might be
involved. The patient cohort should be extensive and well defined, and
deep sequencing and microarray analyses should be used. It's the best
way to go through all the possible pathogens and see which, singly or
combinations, are involved.
Pingback: Inhibition of XMRV by a weapon of mass deamination « LABVIRUS.COM
Interesting post. I have mde a twitter post about this. Others no doubt will like it like I did.
Interesting new insight into HIV immune dysfunction, not just suppression : http://news.yahoo.com/s/nm/20100422/hl_nm/us_hi…
Dysfunction has been s suspected in CFS for years (see Dr. Chia's work on TH1/TH2 imbalance and enteroviruses).
Interesting new insight into HIV immune dysfunction, not just suppression : http://news.yahoo.com/s/nm/20100422/hl_nm/us_hi…
Dysfunction has been s suspected in CFS for years (see Dr. Chia's work on TH1/TH2 imbalance and enteroviruses).