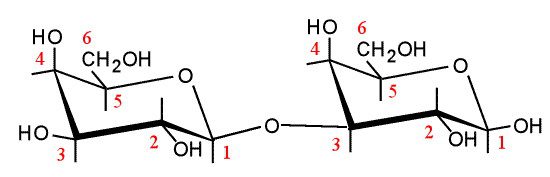

Many natural antibodies are directed against the disaccharide galactose α(1,3)-galactose (α-Gal) (illustration), which is found as a terminal sugar on glycosylated cell surface proteins. Humans, apes, and Old World monkeys lack the gene encoding the enzyme galactosyltransferase, which attaches α-Gal to membrane proteins. Lower primates, most other animals, and bacteria synthesize the disaccharide.
Human serum contains high levels of antibodies specific for α-Gal because the human gut contains bacteria that produce this sugar. Over 2% of serum IgM and IgG antibodies are directed against α-Gal. This antibody binds to the membrane of enveloped viruses that contain α-Gal antigens and triggers the classical complement cascade, leading to lysis of virions and loss of infectivity.
The anti-α-Gal antibody-complement reaction is probably the main reason why humans and higher primates are not infected by enveloped viruses of other animals. Many of these viruses infect human cells efficiently in culture because the complement proteins in serum are inactivated by heating. For example, when vesicular stomatitis virus, human immunodeficiency virus type 2, and human foamy virus are grown in non-human cells which produce α-Gal, the virions can be inactivated by fresh (i.e. not heat-inactivated) human serum. When the same viruses are propagated in human cells, which lack α-Gal, fresh human serum has no effect on viral infectivity. Because of these findings, virus vectors for gene therapy are produced in cells lacking galactosyltransferase to avoid complement-mediated inactivation of the viruses in humans.
When strains of mice that cannot produce any antibodies are infected with vesicular stomatitis virus, titers in the kidney and brain are 10-100 times higher than in normal mice. Furthermore, virus titers in lymph nodes are 10-100 times lower than in antibody-producing mice.
Anti-α-Gal antibodies are an example of how the adaptive immune system cooperates with the innate complement cascade to provide immediate action before specific anti-viral antibodies are developed. Natural antibodies prevent the spread of viruses to vital organs, and improve immunogenicity by enhancing the trapping of antigen in secondary lymphoid organs such as lymph nodes.
Takeuchi Y, Liong SH, Bieniasz PD, Jäger U, Porter CD, Friedman T, McClure MO, & Weiss RA (1997). Sensitization of rhabdo-, lenti-, and spumaviruses to human serum by galactosyl(alpha1-3)galactosylation. Journal of Virology, 71 (8), 6174-8 PMID: 9223512
Ochsenbein AF, Fehr T, Lutz C, Suter M, Brombacher F, Hengartner H, & Zinkernagel RM (1999). Control of early viral and bacterial distribution and disease by natural antibodies. Science (New York, N.Y.), 286 (5447), 2156-9 PMID: 10591647

Pingback: Tweets that mention Natural antibody protects against viral infection -- Topsy.com
flu wasn't mentioned, but is enveloped and has these alpha2-3 and alpha2-6 receptors in HA.
When the natural antibodies kill viruses, do they present them to the specific immune
system, so they can be remembered ? Would make sense
Whoa thanks Vincent. Great post, from my perspective as I have been interested in the antibody response to VSV for quite some time.
I had indeed wondered why, as reported many years ago, humans sera contain natural antibodies with detectable in vitro activity against VSV, whereas sera from my naive mice never did. I am somewhat embarrassed to say I was unaware of the a-Gal differences amongst species.
It's interesting to note that many of us routinely grow VSV in Vero cells (African green monkey) which may lack a-Gal (do they come from OLD WORLD monkeys??), wheras other groups use BHKs which come from rodents and so definitely do not express a-Gal.
Perhaps raising lab mice in a (semi-)sterile environment also has an impact on their natural antibody repertoire?
Also, for those of us working in mouse models, it may be useful to consider the reverse scenario, ie do mice our mice make natural antibodies against antigens from the primate cells we may grow our viruses in? As I say I have not observed this but I have wondered whether mice mount an adaptive antibody response to Vero antigens when immunized with VSV grown in Veros.
Finally, as you mentioned, natural antibodies are generally complement-dependent and also very low titer, at least in the VSV example. It is not obvious to me why this should be the case if they are elicited by an adaptive response to bacterial antigens. Are these bacteria poorly immunogenic? Or do they antibodies just have very low avidity for the a-Gal in the context of a virus particle?
Thanks for a great virologist's virology post!
If natural antibody + complement lyses the virions before they can replicate, there is no adaptive response. If the clearance isn't sufficient, then the interferon response kicks in. If that doesn't work, then antibodies are produced.
African green monkeys are Old World monkeys and hence make a-gal. If you produce VSV in Vero you should use heated serum otherwise your virus titer will be reduced by complement. As for the mice, semi-sterile wouldn't be enough – you would have to raise them gnotobiotically in which case they would have severe digestive problems. I'm not aware of primate antigens incorporated into VSV but I wouldn't be surprised if some were present. The old dogma, that enveloped viruses exclude host proteins when budding, has already been disproven for some viruses. And it's a good question why the a-gal antibodies are such low titer. Your suggestions make sense but I haven't come across the real reason.
Wow. I had not heard that the immune system had such a focus on α-Gal. Have you read how antibodies are able to distinguish between α-Gal and a longer chain of saccharides that terminate with α-Gal? Thanks.
Wow. I had not heard that the immune system had such a focus on α-Gal. Have you read how antibodies are able to distinguish between α-Gal and a longer chain of saccharides that terminate with α-Gal? Thanks.
Pingback: TWiV 82: Immunology in silico
please I have a question ? when new castle disease virus(NDV) is injected in embryonated  eggs it takes several days to kill the chicken embryo and its supposed to kill them  in two days . I ‘ve been told  that these eggs are vaccinated and the virus is neutralized by antibodies .my question is .. if the virus is neutralized by antibodies then why  the chicken embryos died ?and when i made hemagglutination test to estimate the titer of the virus i’ve  found a high titer what is that supposed to mean ?please can you explain that to me  I am ula  from iraq and i really appreciate your help.
Pingback: Natural antibodies | Imageyoo
Pingback: Antibody | Antibody
Pingback: TWiP 97: The seven year itch | This Week in Parasitism